|
Here again is a potential limiting factor for this pathway. Note that the second phosphate group does not require another ATP molecule. The sugar is then phosphorylated by the addition of a second phosphate group, producing 1,3-bisphosphoglycerate. The sixth step in glycolysis (Figure 3) oxidizes the sugar (glyceraldehyde-3-phosphate), extracting high-energy electrons, which are picked up by the electron carrier NAD +, producing NADH. The second half of glycolysis involves phosphorylation without ATP investment (step 6) and produces two NADH and four ATP molecules per glucose. Both of these molecules will proceed through the second half of the pathway, and sufficient energy will be extracted to pay back the two ATP molecules used as an initial investment and produce a profit for the cell of two additional ATP molecules and two even higher-energy NADH molecules.įigure 3. So far, glycolysis has cost the cell two ATP molecules and produced two small, three-carbon sugar molecules. Second Half of Glycolysis (Energy-Releasing Steps) At this point in the pathway, there is a net investment of energy from two ATP molecules in the breakdown of one glucose molecule. Thus, the pathway will continue with two molecules of a single isomer. In the fifth step, an isomerase transforms the dihydroxyacetone-phosphate into its isomer, glyceraldehyde-3-phosphate. The fourth step in glycolysis employs an enzyme, aldolase, to cleave 1,6-bisphosphate into two three-carbon isomers: dihydroxyacetone-phosphate and glyceraldehyde-3-phosphate. The newly added high-energy phosphates further destabilize fructose-1,6-bisphosphate. This is a type of end product inhibition, since ATP is the end product of glucose catabolism. Thus, if there is “sufficient” ATP in the system, the pathway slows down. It is active when the concentration of ADP is high it is less active when ADP levels are low and the concentration of ATP is high. In this pathway, phosphofructokinase is a rate-limiting enzyme. A second ATP molecule donates a high-energy phosphate to fructose-6-phosphate, producing fructose-1,6-bisphosphate. 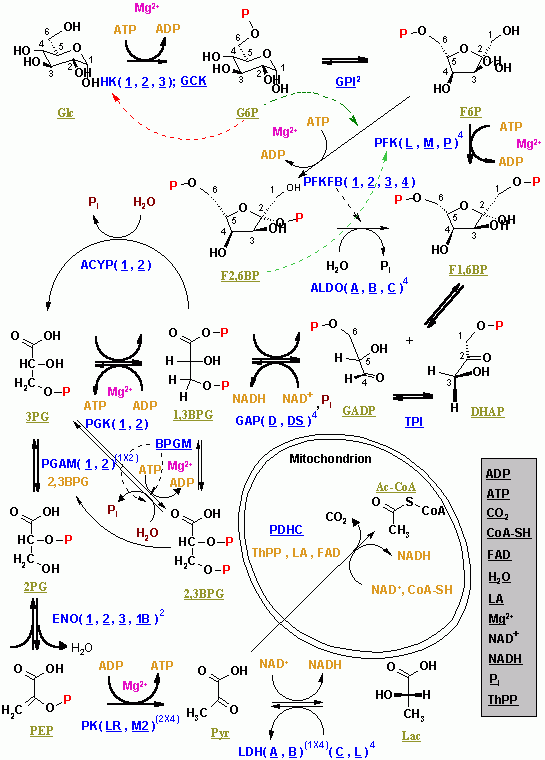
The third step is the phosphorylation of fructose-6-phosphate, catalyzed by the enzyme phosphofructokinase. This change from phosphoglucose to phosphofructose allows the eventual split of the sugar into two three-carbon molecules. An isomerase is an enzyme that catalyzes the conversion of a molecule into one of its isomers. In the second step of glycolysis, an isomerase converts glucose-6-phosphate into one of its isomers, fructose-6-phosphate. This reaction prevents the phosphorylated glucose molecule from continuing to interact with the GLUT proteins, and it can no longer leave the cell because the negatively charged phosphate will not allow it to cross the hydrophobic interior of the plasma membrane. Hexokinase phosphorylates glucose using ATP as the source of the phosphate, producing glucose-6-phosphate, a more reactive form of glucose. The first step in glycolysis is catalyzed by hexokinase, an enzyme with broad specificity that catalyzes the phosphorylation of six-carbon sugars. The first half of glycolysis uses two ATP molecules in the phosphorylation of glucose, which is then split into two three-carbon molecules. Without NAD +, the reaction in step 6 cannot proceed and glycolysis slows or stops. In an environment without oxygen, an alternate pathway (fermentation) can provide the oxidation of NADH to NAD +.Figure 2. However, if there is no oxygen available, NADH is not converted back into NAD +. 
If oxygen is available in the system, the NADH will be converted readily back into NAD + by the later processes in aerobic cellular respiration. If NAD + is not available, the second half of glycolysis slows down or stops. 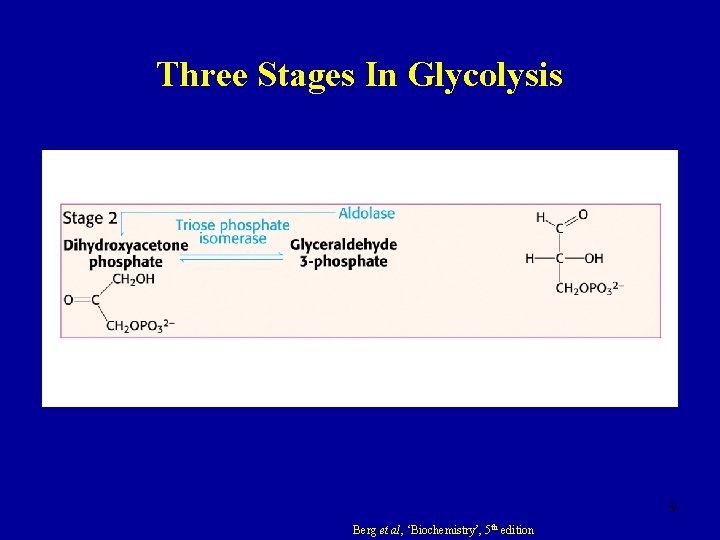
Thus, NADH must be continuously converted back into NAD + in order to keep this step going. However, the continuation of the reaction depends upon the availability of NAD +. NADH contains more energy than NAD +, and is therefore a desired product from this reaction. \), you will notice that during step 6, NAD + is converted into NADH.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
